Report
Ewing GW1*, and Duran JC2
Chief Executive, Mimex Montague Healthcare Limited, Nottingham, England
Medical Director, Department of Integrative Medicine, Hospital San Roque, Las Palmas, Gran Canaria, Spain
Corresponding author
Graham Wilfred Ewing, CEO & Director, Mimex Montague Healthcare Limited, Mulberry House, 6 Vine Farm Close, Cotgrave, Nottingham, United Kingdom, Tel: (0044)-115-9899618, (0044)-7885-755847; Fax: (0044)-115-9899826; E-mail: graham.ewing@mmhcl.co.uk
Received Date: 25th November 2015
Accepted Date: 12th February 2016
Published Date: 16th February 2016
Citation
Ewing GW, Duran JC (2016) A Report of the Ability of Strannik Virtual Scanning to Screen the Health of a Randomly Selected Cohort of Patients. Enliven: Neurol Neurotech 2(1): 001.
Copyright
@ 2016 Dr. Graham Wilfred Ewing. This is an Open Access article published and distributed under the terms of the Creative Commons Attribution License, that permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
There is recognition of the limitations of contemporary biomedicine. Coincidently, there is increasing pressure from governments to explain the
fundamental causes of pathological onset, to move to a preventative healthcare model, to determine the onset of pathologies from their earliest
presymptomatic onset, to integrate different modalities to improve the quality of the treatment and the recovery after treatment, and to reduce the
fundamental cost of healthcare.
This paper reports on a proof of concept study undertaken over the period 2-7th October 2015 in which a randomly selected cohort of patients was
screened using Strannik technology i.e. Strannik Virtual Scanning (SVS). This private study included patients who have their own unique set of medical
conditions and/or medical history including a health psychologist, medical doctors, and medical directors. The reported ailments included patients with
cancer and/or recovering from chemotherapy, cognitive problems, sleep problems, stress, neurological problems, cardiovascular issues, etc.
None of the patients had hitherto been in contact with Mimex Montague Healthcare or hitherto knew of Strannik technology.
Previous data from Russian clinicians indicated that SVS was able to diagnose typically 2-23% more accurately that the range of diagnostic tests against
which it was compared and which was being routinely used in their Russian medical clinics.
This brief study – reporting initially on the outcome of SVS to rapidly screen a group of patients – indicated that SVS was able to define the complete
health of each patient and to make what appears to be an accurate report of their health. In 17 of the 20 patients the SVS results coincided with the
patient’s declared medical conditions. In 2 cases the patient’s health history was not declared and in 1 case the patient was unable to complete the test
in a satisfactory manner. This encouraging report is consistent with and supports the claims of various clinicians, doctors and researchers in the Russian market. It is emphasised that the conclusions from this initial study needs be supported by a double-blind clinical study involving a statistically significant patient cohort
Keywords
Strannik; Mathematical model; Human brain project; Strannik virtual scanning
Abbreviations
SVS: Strannik Virtual Scanning
Introduction
Strannik technology [1] is based upon an original mathematical model of the autonomic nervous system and physiological systems. It meets the specification defined by the European Commission’s Human Brain Project [2] for a new generation of medical technology, based upon cognitive input, which can determine the health of the patient (in particular the complex pathological correlates of Alzheimer’s disease); which recognises what is the function of the brain – what does it do and how does it do it; and which is able to recognise and adapt an understanding of the multi-level function of the brain to improve/treat patient health [3].
Strannik Virtual Scanning (SVS) is the first medical technology which is able to provide a comprehensive screen of the health of the patient including the genetic and phenotypic components [4] of most common pathologies.
Strannik Virtual Scanning
An SVS [5,8] test enables the practitioner to understand the health of the patient at different levels of significance e.g.
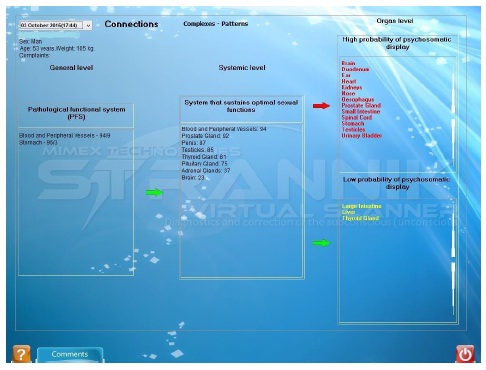
• Figure 2: At the Systemic level and Organ level and to understand those areas where pathological development will occur in future (see ‘high probability of psychosomatic display’).
The example if that of a male patient in which their most deviated physiological system is that of sexual function involving blood and peripheral blood vessels, the sexual organs, endocrine glands and the brain.
• Figure 4: At the Cellular level where (i) a system of coloured markers is used to identify cellular change in each organ and (ii) illustrate the morphological situation of each organ in terms of processes which support the development of connective tissue, functions acceleration and new cell formation.
• Figures 5-8: At the pathological level where it is possible to identify the specific pathologies in each organ and to identify the genetic and phenotypic components of each pathology.
The examples are of patients with (i) calculous prostatitis (figure 5), (ii)with a dementia-type disorder (see encephalopathy: figure 6), (iii) spinal problems (figure 7), and (iv) diabetes (figure 8). Indices in blue are indicative of genotype, indices in red are indicative of phenotype, 7-10 units is the boundary between presymptomatic and symptomatic i.e. if below 7 units the pathology is manifest at its presymptomatic stage and from its presymptomatic onset whilst if above 7-10 units the pathology is manifest at its symptomatic stage.
In addition the technology incorporates a number of cross-checks which enable the practitioner to confirm the nature of the pathology and avoid mis-diagnosis e.g.
• Figure 1: Is a summary report which indicates the presence of processes (i) which stimulate the hormone-related growth of new cells, (ii) whether new cell growth is being inhibited or sustained, (iii) whether there is carcinogenic influence upon the genetic system of core cells, and finally (iv) the organs which are no longer able to influence or control metatic activity.
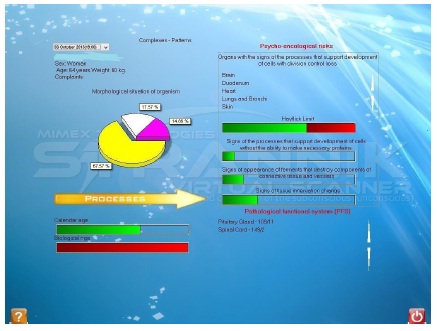
• Figure 3: Looks at (i) the morphological situation of the organism in terms of new cell growth, old cells, functions deceleration, processes which support the development of connective tissue; (ii) a comparison of the patient’s calendar age with their calculated biological age; (iii) the specific organs which exhibit signs of the processes which are potentially cancer-forming and malignant i.e. which support the development of new cells with loss of control of cell division [6]; (iv) Hayflick [7] limit violations and other processes which may influence the function of the organism e.g. signs of fermentation processes which may destroy connective tissue and vessels; and (v) the specific organs which are functioning as part of a pathological functional system.
Study Protocol
The patients were introduced into the test room where they were weighed. They completed the registration form which is a necessary part of the test procedure. This included their personal identifying details which could be their name or an identifying code, weight, birth date and gender. There was no disclosure of their personal health prior to undertaking the test.
Each patient was given a demonstration of the SVS test which involves studying a video for 15 seconds (and memorising the colours in the video) and then, following the imposition of a colour filter, using the ‘mouse’ to select colours from a colour palette which could be added or subtracted from the modified video until they completed the task of recovering to the best of their ability the colour balance in the original video. See demonstration video www.montaguehealthcare.co.uk/presentation.php The patient was coached for a period, typically 3-7 videos, to understand how the test works before finally being asked to undertake the SVS test. This was repeated 4 or 5 times with different video sequences to gather the cognitive data required by the test patient. Finally the data was processed by the mathematics of the program to provide the SVS test report.
The test was straightford for most patients,the process taking typically 15-30 minutes, but for those with severe and/or problematic neurological or cancerous conditions the process took typically 45-60 minutes.
This article focuses upon (i) the ability of SVS to rapidly and in expensively screen the health of a cohort of 20 randomly selected patients with a wide range of complex medical conditions e.g. diabetes, cardiovascular indications, cancers, etc. As outlined, the test method adhered to the basic protocol outlined in the Strannik Operating Manual [8]. As the technology was considered to be completely non-invasive, and without any possible risk to patient health, and testing was undertaken in a non-clinical location, it was not considered to be necessary to secure ethics committee approval. Although the study was not entitled a 'double-blind clinical study' the patients were not familiar with the test procedure and could not possibly falsify their test results i.e. it was almost identical to the protocol which would be used in any double-blind clinical studies. Similarly the practitioner was completely unaware of the health of each patient until after the test had been completed and could not therefore falsify the test results. In addition, the patient population included three medical doctors, a medical director, nutritionist, medical student and psychologist and was overseen by a medical doctor/specialist in Integrative Medicine.
The Strannik practitioner reported the major medical indications which were identified by the test procedure. The patients reported the medical conditions which were known to them i.e. which had been discussed with their doctor(s). The results were subsequently reported (or were already known) to the supervising doctor who is the co-author of this report.
Patient Reports
The following patient reports will focus upon the key features which were identified in the SVS report.The reported indices are of rate of protein reaction rather than levels of biochemical markers.
Note 1: pathologies are reported in a ratio of phenotype/genotype e.g. 5/14.
Patient 1: Male, 54 years, 78 kgs
Patient history reported by the patient included: Urticaria, Cystitis (arising from bacterial infection of the prostate), kidney stones (eliminated using lemon juice and olive oil), diarrhoea
SVS report identified: Urticaria, Cystitis, Stomach Ulcer 5/14, and Gastritis
Patient 2: Female, 38 years, 59 kgs
Patient reported: Rosacea, haemorrhoids, and of problems with her digestion, kidneys and nervous system.
SVS report identified: Psoriasis, haemorrhoids, a fungal infection or other similar adverse influence upon intestinal flora, and indication of hepatocirrhosis (Note 1)
Patient 3: Male, 53 years, 105 kgs
Patient reported: Patient reported consequences of severe neck trauma (fused C4 and C5), problems of digestion, severe stress, lack of exercise in the preceding 9 months, occasional light-headedness (avoiding driving).
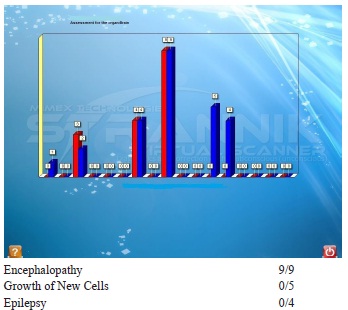
SVS test identified: (i) Most destabilised system: system which maintains optimal sexual function. (ii) Patient undersevere stress influencing brain function including encephalopathy 9/9, spinal arachnoiditis 15/5, prostatis, type 2 diabetes 14/4, cardiac insufficiency 25/0, stomach ulcer 36/0. (iii) New cell growth in a number of organs but which do not have oncological significance at this stage.
Patient 4: Female, 64 years, 60 kgs
Patient reported: n/a
SVS test identified: (i) Major Hayflick violation. (ii) Patient with severe problems of brain function and with dysfunction of spinal cord and adrenal glands, probably contributing to poor quality sleep. (iii) Most deviated system was that of ‘posture’. This was supported by indications of arthritis, radiculitis and osteoporosis. (iv) Indications of growth of new cells and breathing problems (as chronic breathing insufficiency) in the lungs.
Patient 5: Female, 67 years, 64 kgs
Patient reported: Sleeping problems, hypertension, anxiety/depression
SVS report identified: (i) Most deviated system: posture. (ii) A range of stress-related markers including pathological onset in the spinal cord, adrenal insufficiency, pyelonephritis, colitis and proctitis in the large intestine, osteochondrosis 16/6, hypothyrosis and thyrotoxicosis in the thyroid.
Patient 6: Male, 58 years, 62 kgs
Patient reported: Osteoporosis, cervical and lumber stenosis, stress, cold hands and feet, sinusitis
SVS report identified: (i) Major Hayflick violation. (ii) Most destabilised system is that of Sleeping pattern. (iii) Osteoporosis 14/12, impaired spinal circulation 27/10, spinal arachnoiditis 12/10, frontitis 4/4. (iv) New cell growth in a number of organs but which do not have oncological significance at this stage. (v) adrenal insufficiency 39/38.
Patient 7: Female, 67 years, 77 kgs
Patient reported: n/a
SVS report identified: Indications of cancer development (Hayflick violation), diabetes 19/11, neuritis, gastritis, asthma.
Patient 8: Female, 38 years, 50 kgs
Patient reported: Mitral valve issue experienced whilst training for triathlons several years previously. Recently gave birth to a baby.
SVS report identified: Preindications of cancer (increased Hayflick limit), myocardial dystrophy 20/23, and cardiac insufficiency 18/14.
Patient 9: Female, 39 years, 75 kgs
Patient reported: Lymphatic problem i.e. swelling of the lymph nodes.
SVS report identified: Patient with severe problems of brain function, preindications of cancer (increased Hayflick limit), insufficiency of the adrenal cortex 12/0, diabetes 12/13, idiopathic hypotension 13/17.
Patient 10: Male, 59 years, 78 kgs
Patient reported: Nearing completion of his chemotherapy course following treatment for lung cancer. Experienced pulmonary embolism during chemotherapy and currently being prescribed heparin. High blood pressure, history of type 2 diabetes in the family, urticaria.
SVS report identified: Patient with significant indications of cancer (elevated Hayflick limit), residues of new cell growth in the lungs, insufficiency of the adrenal cortex 17/0, duodenal ulcer 10/0, dyskinesia in the small intestines, cardiac myopathy 11/9, new cell growth in a number of organs but which do not have oncological significance at this stage, urticaria 5/5 and dermatitis 7/9.
Patient 11: Female, 56 years, 86 kgs
Patient reported: No significant health issues, stressed following husband’s stroke
SVS report identified: No significant health issues. Biological age younger than her calendar age.
Patient 12: Male, 64 years, 80 kgs
Patient reported: Unable to speak following a stroke
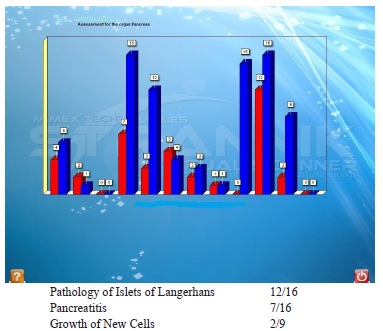
SVS report identified: Patient with severe problems of brain function, indications of new cell growth (elevated hayflick limit) in various organs e.g. pancreas and prostate, epilepsy, prostatitis, pyelonephritis 23/4 and renal insufficiency 9/7, major diabetes signal 64/23, neuritis 17/1, ganglioradiculitis 20/0.
Patient 13: Female, 52 years, 50 kgs
Patient reported: Recovering from chemotherapy following lung cancer, sleeps for 2-4 hours per night, tendonitis in shoulder, dermatitis and diabetes.
SVS report identified: Duodenal ulcer 10/21, duodenitis 10/13, rhinitis (perhaps the consequence of immunosuppression following chemotherapy 0/24), diabetes 12/16, impaired spinal circulation.
Patient 14: Male, 54 years, 80 kgs
Patient reported: Previous cardiac arrhythmia (4-5 years previously)
SVS report identified: Little of any significance
Patient 15: Male, 50 years, 90 kgs
Patient reported: Raised blood pressure due to work-related stress
SVS report identified: Insufficiency of adrenal cortex, presymptomatic indications of urticaria and neurodermatitis, growth of new cells (elevated hayflick limit) in the testicles, hypothyrosis.
Patient 16: Male, 45 years, 89 kgs
Patient reported: sleep problem, nose problem
SVS report identified: (i) most destabilised system: body temperature. (ii) rhinitis, myelitis, neurodermatitis, prostatitis, cardiac insufficiency 10/12, ischaemic heart disease 9/7
Patient 17: Female, Nutritionist, 46 years, 74 kgs.
Patient reported: n/a
SVS identified: Patient with problems of brain function, overweight and weight related issues to heart function (cardiac myopathy 6/18 and myocardial dystrophy 7/12) and diabetes 11/13
Patient 18: Female, 31 years, 54 kgs
Patient reported: Rheumatoid arthritis
SVS identified: Patient with problems of brain function difficult test/diagnosis. Patient was unable to complete the test in a satisfactory manner. Tested on three occasions. Elevated Hayflick limit.
Patient 19: Male, 50 years, 65 kgs
Patient reported: insomnia. Has not slept for the last 20 years - takes 3 sleeping pills each evening. Patient is clearly alarmed at their current state of health.
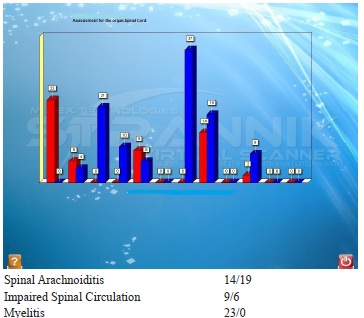
SVS identified: Patient with problems of brain function difficult test/diagnosis although not as bad as expected.Tested three times. Test results unsatisfactory. Indications of cancer (elevated Hayflick limit). Problems with spinal cord (spinal arachnoiditis 14/19 and impaired cerebral circulation 9/6) and hence of problems with sleeping pattern.
Patient 20: Male, Medical Student, 28 years, 70 kgs
Patient reported: Under heavy stress due to intense preparations for forthcoming examinations, neurodermatitis.
SVS identified: Minor Hayflick violation. Neurodermatitis, latent herpes, otherwise healthy.
Summary
The SVS test results are interesting because they were conducted in an entirely blinded fashion in which the Strannik practitioner was completely unaware of the health of each patient yet provided, what appears to be, an accurate summary of the health of each patient. This compares with the medical diagnosis conducted by the primary care GP whereby he has the benefit of (i) knowing the patient's medical history, (ii) being able to visually assess the patient, (iii) ask the patient to describe their medical ailment, symptoms and associated information, and (iv) conduct additional tests to verify their medical conclusions.
The report identified a wide range of medical conditions in each patient of which the most prominent and/or significant are summarised in this report. Of the 20 patients tested the test report the majority of the patients (17) had their reported medical indications confirmed by SVS i.e. the SVS test results were consistent with their known medical conditions. Two of the patients did not provide information about their health history therefore, in these two patients, it was not possible to conclude whether the test results were correct or not.
The 17 patients provided circa 27 comments about their health i.e. their known medical history. The SVS test gave information on more than 65 medical indications which appeared to be consistent with their declared medical history and which was known to the medical team monitoring this study. Apart from the two patients who did not declare their medical conditions one person was cognitively hindered and was not able to complete the test in a satisfactory manner. There were no medical indications declared by the patients which were not be confirmed by the SVS test.
Note 2: The SVS test results identified a number of medical conditions which may be associated with environmental conditions in Gran Canaria e.g. urticaria, pyelonephritis, and hepatocirrhosis.
A significant number of the patients were identified with indications of new cell growth (patients 3,4,6,9,10,12,13,18,19) however none of the patients were positively identified with a malignancy. Their conditions were clearly of concern. Cancer generating processes were clearly identified e.g. following chemotherapy, but were considered to be benign or at a presymptomatic stage of onset.
A number of patients with no medical problems, minor medical problems were introduced for comparative purposes. The test was able to determine whether they were completely healthy and/or the nature of their minor ailments.
This report illustrates the potential of the Strannik technology, in particular of the SVS test, to support the work of the doctor e.g. to make an immediate and reasonably accurate assessment of patient health without need for further testing in the secondary care environment, to use this information to improve patient lifestyles, and/or to recommend therapeutic interventions and/or practices which could improve their quality of life. It meets the requirements of independent institutes e.g. the UK’s National Institute of Health & Care Excellence (NICE), for ‘plausible promise’ and hence that further testing e.g. in a double-blind clinical study, should be undertaken in order to fully evaluate the potential of this technology to assist the doctor to reduce the complexity and hence the cost of medical testing.
References
- Ewing GW, Ewing EN (2008) Cognition, the Autonomic Nervous System and the Physiological Systems. J Biogenic Amines22: 140-163.
- Ewing GW, Grakov IG (2015) A Comparison of the Aims and Objectives of the Human Brain Project with Grakov’s Mathematical Model of the Autonomic Nervous System (Strannik Technology). Enliven: Neurol Neurotech 1: 002.
- Ewing GW (2009) A Theoretical Framework for Photosensitivity: Evidence of Systemic Regulation. J Comp Sci Syst Biol2: 287-297.
- Ewing GW (2013) A Comparison of the Diagnostic Scope of Biomarker techniques, Genetic Screening and Virtual Scanning. Immun Endocrine Metabol Agents Med Chem13: 35-45.
- Ewing G, Ewing E, Hankey A (2007) Virtual scanning--a new system of medical assessment and treatment: Part I. Assessment. J Altern Complement Med13: 271-286.
- Winter C, Kristiansen G, Kersting S, Roy J, Aust D, et al. (2012) Google goes cancer: Improving outcome prediction for cancer patients by Network-Based ranking of marker genes. PLoS Comput Biol 8: e1002511.
- Shay JW, Wright WE (2000) Hayflick, his limit, and cellular ageing. Nat Rev Mol Cell Biol1: 72-76.
- www.montaguehealthcare.co.uk/OperatingManual.pdf